Trial 1
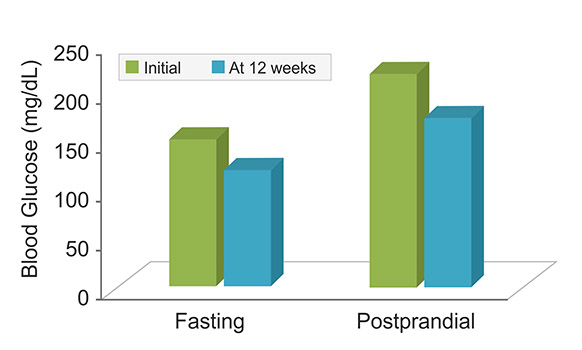
A multicenter, flexible dose, 12-week, open trial involving newly-diagnosed type 2 (NIDDM) patients was
carried to assess efficacy of Pterocarpus extract. Patients were treated with increasing doses of Pterocarpus
extract (i.e. 2 g, 3 g and 4 g at a gap of 4 weeks each). At the end of 12 weeks, 69% patients showed
significant control of blood glucose (both fasting and postprandial levels).
Additionally, mean HbA1c was found to be decreased significantly (P < 0.001) at the end of the trial. There were no side-effects reported during the study period. In conclusion, Pterocarpus extract may be helpful in the treatment of newly-diagnosed or untreated mild NIDDM patients (ICMR, 1998).
Trial 2
In another 36-week, randomized, double-blind, multicenter, flexible dose trial, blood lowering effect of dried
aqueous extract of the heartwood of P. marsupium was compared with that of tolbutamide, a standard
antidiabetic agent, in newly-diagnosed or untreated type 2 diabetic patients.
Patients were randomly divided into test group (n=172) and tolbutamide group (n=177) and were treated with
increasing dosage (i.e. Vijaysar: 2 g, 3 g and 4 g/day and Tolbutamide: 0.75 g, 1 g and 1.5 g/day) at a gap of 4
weeks each. Results demonstrated that treatment with Vijaysar was able to lower blood glucose level in 86%
patients, which was comparable to tolbutamide efficacy (i.e. 94%). Hence, it was concluded that Vijaysar could
be an effective blood glucose lowering agent in type 2 diabetes without any significant side effects (ICMR,
2005).
|
||||||||||||||||||||||||||||||||||||||||||||||||